Our Approach
Pioneering the next generation of drug discovery
Creating new therapies by treating cellular mechanisms that cause disease
Overview
Hundreds of diseases remain unsolved because they haven't been addressed in their cellular context.
Many processes like signaling, transcription, trafficking and protein interactions involve the orchestration of hundreds of proteins within the cell. Understanding and treating the dysfunction of these mechanisms at the cellular level is often the key to therapeutic success. Rather than traditional discovery approaches that consider a purified target outside this cellular context, Octant engineers cellular sensors to report on the processes we are targeting. We use these cellular sensors to better understand how to modulate these processes and to develop new drugs against them.
The Octant Navigator is a next-generation approach to drug discovery
The Octant Navigator accelerates design, build, test, learn iterations across chemistry and biology to build drugs against cellular mechanisms.

Generative Biology
Generative Biology is our high-throughput approach to drug design through understanding the cellular mechanisms that underlie disease. We build mammalian synthetic biology toolkits to rapidly design, build, and test cellular sensors that report on activities like signaling, expression, trafficking, stability and interactions of disease target pathways. Using RNA-barcoding technologies, we can simultaneously read out on hundreds to millions of conditions to build drugs with highly desired profiles.
Generative Chemistry
Our nano-scale automated chemistry synthesis platform enables us to build large libraries of related molecules to engineer quality drug candidates. Unlike approaches that use large libraries of unrelated compounds, we empirically screen thousands of analogs to discover which chemical modifications turn weak target binders into drug-like compounds. By screening focused libraries in a direct-to-biology approach we can fine-tune pharmacologies to satisfy challenging target profiles using iterative learning loops between synthesis and screening.

Generative AI
Advanced machine learning and automation technologies enable Generative Biology and Generative Chemistry to iterate in a positive feedback loop. We design, build, test, and learn weekly, iterating on the results of tens of thousands of novel chemicals screened against next-generation assays. Our computational pipeline shapes these millions of empirical readouts into actionable insights quickly, enabling us to design better molecules.
Deep Mutational Scanning
A new frontier for understanding protein function
Deep Mutational Scanning (DMS) harnesses Octant’s Generative Biology platform to design, build, and screen thousands to millions of cell lines, generating unique insights into a protein’s function. We engineer a distinctly barcoded cell line for every amino acid substitution of the target, mapping the functional response molecules acting on each of those cells. Precisely understanding the extent to which a residue or mutation of a protein differentially drives each of its activity pathways opens entirely new capabilities for building better drugs.
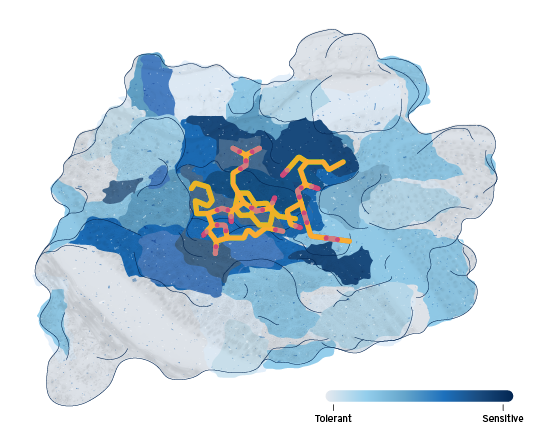
Our therapeutic approach
The Octant Navigator enables new approaches to treat serious diseases.

